-
PROIZVODI
- Laboratorijski instrumenti
-
Laboratorijska oprema i opskrba
Opći potrošni materijal laboratorija InstrumentiUređaj
- Cijevi
- Dispenzeri i Droperi
- Grab Uzorkivači
- Kleme, Prstenovi i Stalci
- Ostale Aparature
- Pipete
- Pomagala za Pipete
- Posudice
- Posudice i Zdjelice
- Pribor za Vaganje
- Stalci
- Četke
- Štapići za Miješanje
Stakleno/plastično posuđe - Automatizirani laboratorijski sustavi
- Kemija, reagensi i standardi
- Mikrobiologija
- Laboratorijski mjerači i sonde
-
Online analizatori
EZ Series Analysers
- Iron
- Aluminium
- Manganese
- Phosphate
- Chloride
- Cyanide
- Fluoride
- Sulphate
- Sulphide
- Arsenic
- Chromium
- Copper
- Nickel
- Zinc
- Ammonium
- Total Nitrogen
- Total Phosphorus
- Phenol
- Volatile Fatty Acids
- Alkalinity
- ATP
- Hardness
- Toxicity
- Sample Preconditioning
- Boron
- Colour
- Nitrate
- Nitrite
- Silica
- Hydrogen Peroxide
- EZ Series Reagents
- Online senzori i kontroleri
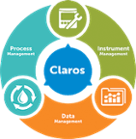
- Claros Water Intelligence System
- Testni kompleti i trake
- Uzorkovanje
- PARAMETRI
-
Softverska rješenja
-
Claros Sustav za inteligentno upravljanje vodom
Proizvodni stupovi Process Management
- Namjena rješenja:
- Otklanjanje BPK (BOD)/KPK (COD)
- Nitrifikacija/denitrifikacija
- Uklanjanje fosfora
- Upravljanje muljem
Data Management- Namjena rješenja:
- Prikupljanje
- Vizualizacija i analitika
- Izvještavanje
- Točnost podataka
Instrument Management- Namjena rješenja:
- Održavanje
- Rješavanje problema
- Udaljeni pristup
- Usporedba vrijednosti iz laboratorija i postupka
-
Claros Sustav za inteligentno upravljanje vodom
- Industrija
- PODRŠKA
- NOVOSTI I DOGAĐAJI
Find the best solution for each drinking water parameter
Turbidity is one of the most important parameters in the drinking water treatment process. By monitoring turbidity in various parts of the treatment process, you can ensure regulatory compliance and have confidence in the quality of your water.
Turbidity is the relative clarity of a solution. Clarity is decreased by suspended solids such as clay, algae, organic matter or microorganisms. When light shines through the solution, these particles scatter and absorb the light. Turbidity is measured by the light reflected off of these particles at a 90 degree angle.
Given the wide range turbidity in the various steps of the treatment process, it is important to select the right instrument for each application.
| Turbidity Range | >10 NTU (bis zu 999 NTU) | <10 NTU |
| Turbidity Application |
Raw influent water
Clarifier effluent
Filter backwash water |
Filter effluent Combined filter effluent |
| Turbidity Solution | ||
pH is another important parameter to measure and control in a drinking water facility. pH directly impacts the degree of coagulation and flocculation that remove total organic carbon from raw inlet water. pH also affects the disinfecting power of chlorine and therefore must be kept in a very narrow range (7.0-7.8 pH) during and after the disinfection process; this range maximises the effectiveness of the disinfectant (which is less effective in pH > 7.8) while minimising corrosion of systems
caused by low pH (< 7.0).
Disinfection with chlorine is often a preferred method to kill pathogens in drinking water. When chlorine is added to water, it forms hypochlorous acid (HOCl) which is a very strong disinfectant. This acid further dissociates to hydrogen and hypochlorite ions (H+ and OCl-), a significantly weaker disinfectant. The percentage of hypochlorous acid is higher in acidic environment (pH<7.5) and lower in basic environment (pH>7.5). Therefore, the same amount of chlorine added will have different
disinfecting power based on the pH of the water. This balance is further affected by temperature.
This sensitive balance of chlorine chemistry in water can create various challenges for drinking water plants that want to make sure they have added enough chlorine to ensure water safety, but not too much to affect water taste or increase the chance of chlorine reacting with natural organic matter to form cancerogenic disinfection byproducts.
There are two main methods for measuring chlorine levels in water, and choosing the right one for your application depends on various factors
| Chlorine measurement method |
Amperometric | Colorimetric |
| Main benefit |
Ideal for process control with fast reaction to chlorine
concentration change. |
High accuracy without calibration. |
| Best suited for | Stable pH, temperature and flow. | Any application with changing sample characteristic (pH, temperature, flow). |
| Chlorine analyser and main features |
Sensor specificity to HOCl. No sanitary drain required 9184 sc Total Free Chlorine (TFC) amperometric analyser |
Not affected by process changes and no calibration required. Low maintenance cost thanks to 30 day unattended run time CL17 Analyser for total chlorine |
| Eliminates need for external buffer. Optional pH probe. No sanitary drain required CLF10 sc Free chlorine analyser, pHD sensor, metric |
Organics
Natural organic matter (humic, fulvic, tannic acids, etc.) may be present in natural water sources, and one of the main goals of the drinking water treatment process is to remove these dissolved
organics. This is especially important when chlorine is used as a disinfectant, as chlorine reacts with organics to form carcinogenic disinfection by-products (THMs, HAAS, etc.) Dissolved organic material are monitored by 254 nm UV absorption.
Nitrites
Nitrates are usually found in high concentrations in ground water, especially when activities near the well can potentially contaminate the water supply. Nitrates are also found in surface water
when nitrate fertilisers are leached during excessive rainfalls.
Sludge monitoring
Sludge thickening reduces the volume collected from the clarifier and sludge, dewatering the weight by centrifugation or filtration. Both of these processes can be optmised using online turbidimeters that measure suspended solids.